
Ostwald in particular advocated the view that thermodynamics dealt only with energy and how it is transformed in the everyday world. They argued that the laws of thermodynamics need not be based on mechanics, which dictated the existence of invisible atoms in motion.
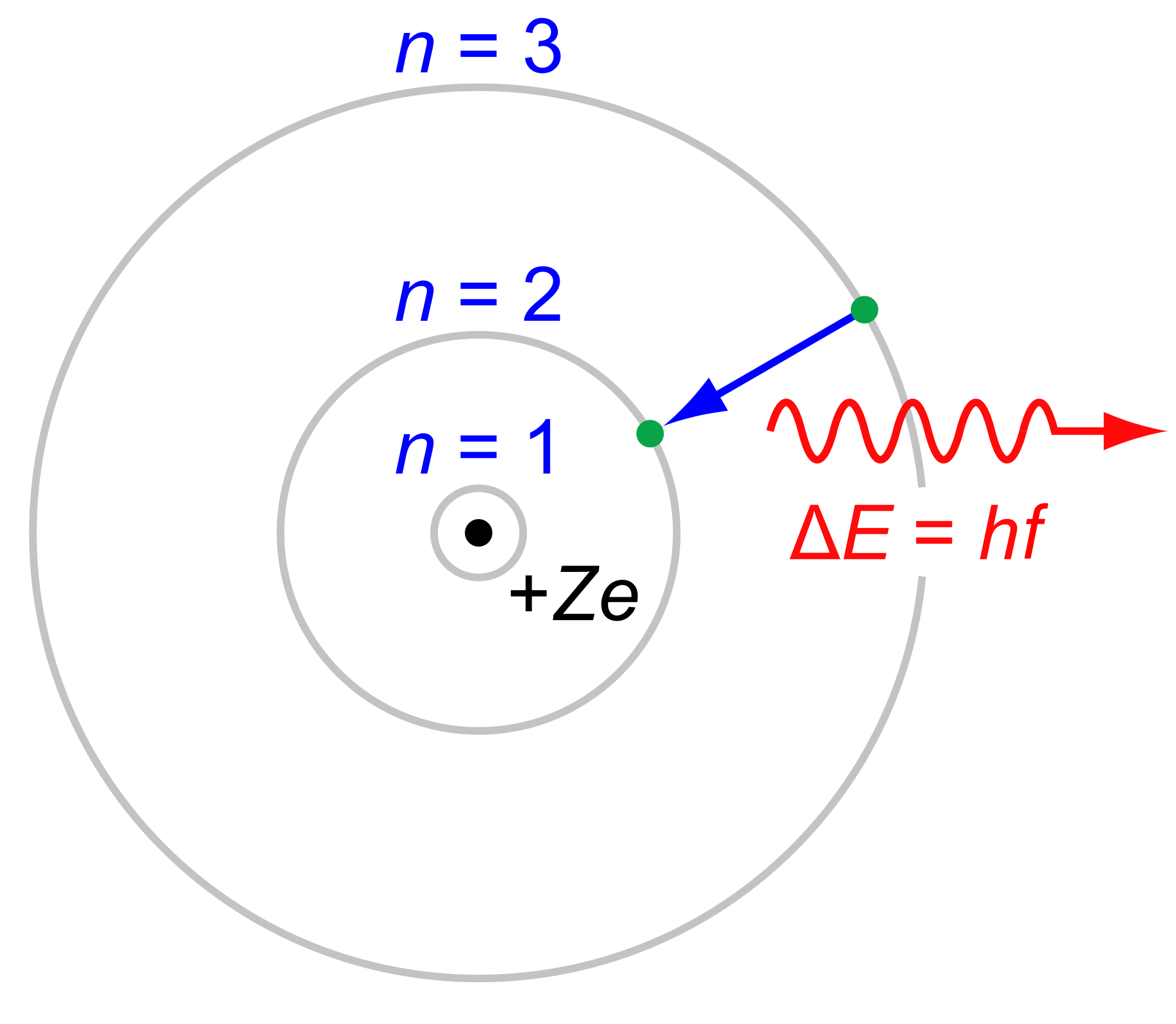
Ernst Mach and the physical chemist Wilhelm Ostwald were among those who chose to deny their existence. When Einstein's paper first appeared in 1905, the notion of atoms and molecules was still a subject of heated scientific debate. Einstein explained the motion in detail, accurately predicting the irregular, random motions of the particles, which could be directly observed under a microscope. Einstein then reasoned that if tiny but visible particles were suspended in a liquid, the invisible atoms in the liquid would bombard the suspended particles and cause them to jiggle. In 1827, the English botanist Robert Brown noticed that pollen seeds suspended in water moved in an irregular "swarming" motion. In a separate paper, he applied the molecular theory of heat to liquids to explain the puzzle of so-called "Brownian motion". He used mechanics, atoms and statistical arguments to formulate a "general molecular theory of heat." Einstein developed this statistical molecular theory of liquids for his doctoral dissertation at the University of Zurich. Įinstein was supposedly unaware of Boltzmann's work when he began independently deriving the second law of thermodynamics from 1902 to 1904 to develop his own form of statistical mechanics. The pair finally married in 1903 after Einstein got his job at the Patent Office. Einstein's family opposed any talk of marriage, even after Mileva gave birth to a daughter (who was apparently given up for adoption).
While a student at the Zurich Polytechnic Institute, Einstein met a young Serbian woman, Mileva Maric, the only woman in his physics class. The statistical improbability of those molecules doing so, however, was the source of the seeming irreversibility observed in nature. He reasoned that there are so many atoms and molecules that make up something as small as an ice cube, for example, that it is extremely unlikely-although not impossible-for the molecules in a melted ice cube to return from the disorder of a liquid to their original orderly arrangement. This arose from the second law of thermodynamics, which dictates that most natural processes are irreversible, in seeming contradiction to the Newtonian mechanics of atoms.īolztmann reinterpreted the second law as statistical, rather than absolute. Along with the American physicist J.Willard Gibbs, Ludwig Boltzmann used the kinetic theory to resolve the so-called "reversibility paradox" in physics. In the 19 th century, physicists had refined the kinetic theory of gases, which described heat as an effect of the nonstop agitated motion of atoms. This time, his subject was the kinetic theory of gases, but the paper was equally groundbreaking in its conclusions. In May the journal received another paper from Einstein. It was published in the Annalen der Physik. In March of 1905, a young patent clerk in Switzerland named Albert Einstein submitted a groundbreaking paper extending Planck's 1900 notion of quanta to the wave/particle dual nature of light. Photo Credit: American Institute of PhysicsĮinstein and his first wife with their first-born son.
